Reconstructive lymphatic surgery: what’s the deal?
“Are you telling me I have to wear these compression stockings my whole life? Can’t I have surgery?”
It’s a common question we hear in our practice. In a world where surgical techniques are developing so fast it’s often difficult to explain to a patient with lymphedema that the first choice of treatment is the conservative treatment, i.e. decongestive lymphatic therapy (DLT).1,2 It consist of two phases. In the first phase we want to reduce the swelling as much as possible by applying multilayer bandaging in combination with skin care, exercises and optionally manual lymph drainage. The purpose of the second phase is to maintain the results obtained in the first phase with a compression garment. Exercises, skin care and manual lymph drainage (if needed) are continued.2
“So, there are no surgical techniques for the treatment of lymphedema?”
Actually, there are. Two types of reconstructive surgical techniques exist for the treatment of lymphedema: lymphovenous anastomosis and lymph node transfer. The lymphovenous anastomosis (LVA) was first described in the sixties.3,4,5,6 The idea behind the LVA is to create a physiological bypass so that lymph fluid doesn’t have to pass the area of obstruction. By making a connection between a functional lymphatic vessel and the venous system at capillary level, the lymph fluid can be transported from the lymphatic channels into the venous system.7,8 For this type of surgery the presence and quality of functional lymphatic vessels are important. If these are not appropriate, a lymph node transfer could still be an option.9
Lymph node transfer (LNT) is a more recent procedure and was first clinically used in the eighties.10 Healthy vascularized lymph nodes are transferred from a donor site to the recipient side on the affected limb. The decision of the location of the donor site depends on different factors such as availability of lymph nodes, morbidity, etc.11 The theory behind this type of reconstructive surgery is that it would promote lymphangiogenesis12 i.e. the growth of lymphatic vessels out of pre-existing lymphatics13. Another hypothesis is that the transferred nodes work like an internal pump absorbing the fluid into the flap which is then transported to the venous system by a suction mechanism.14 When the recipient side is prepared, it’s also important to perform a wide excision of the scar tissue which can be present as a consequence of previous surgery and radiation therapy. Otherwise, the scar tissue could cause venous constriction.9 In some cases LVA and LNT can also be combined.
“They sound like good treatments. Why aren’t they a part of the standard treatment?”
Despite these kind of surgeries are already being executed, the high-quality scientific evidence is limited.
Recent systematic reviews about LVA discuss that there is often a lack of long-term follow-up15, no control groups, small sample sizes, insufficient outcome reporting16, heterogeneity among reported outcomes and risk of selective reporting bias17. To our knowledge, no randomized controlled trials have been performed on the effect of LVA in patients with lymphedema. Randomized multicenter controlled trials with greater sample sizes are needed to draw firm conclusions and to improve treatment.17,18
About the results of LNT, only one randomized controlled trial exists. In this study 36 patients with stage II arm lymphedema after breast cancer were included. Eighteen women received a vascularized LNT followed by manual lymph drainage the first month and wearing a compression garment for the next five months. Additionally, they had to perform daily a gentle pumping pressure onto the flap during the first three months after surgery. Six months after LNT, conservative treatment was discontinued for twelve months. The other eighteen women only received conservative treatment (manual lymph drainage and compression garment) for six months which was thereafter also discontinued for twelve months. Patients were evaluated at start and after eighteen months of follow-up. The mean volume reduction was significantly larger in the group who received the LNT (57 % versus 18 %, p = 0.000) than in the group without the surgery. The women who received LNT also had significantly fewer infections, greater reduction of pain and feelings of heaviness and showed significantly better functional improvement.19 This looks promising, however, randomized controlled trials with greater sample size are necessary to confirm these positive results. Other studies performed on the effect of LNT had a lower level of evidence.
“So we need in the future new high-quality research to know if the current applied surgeries are effective and can be continued. Are there any prospects?”
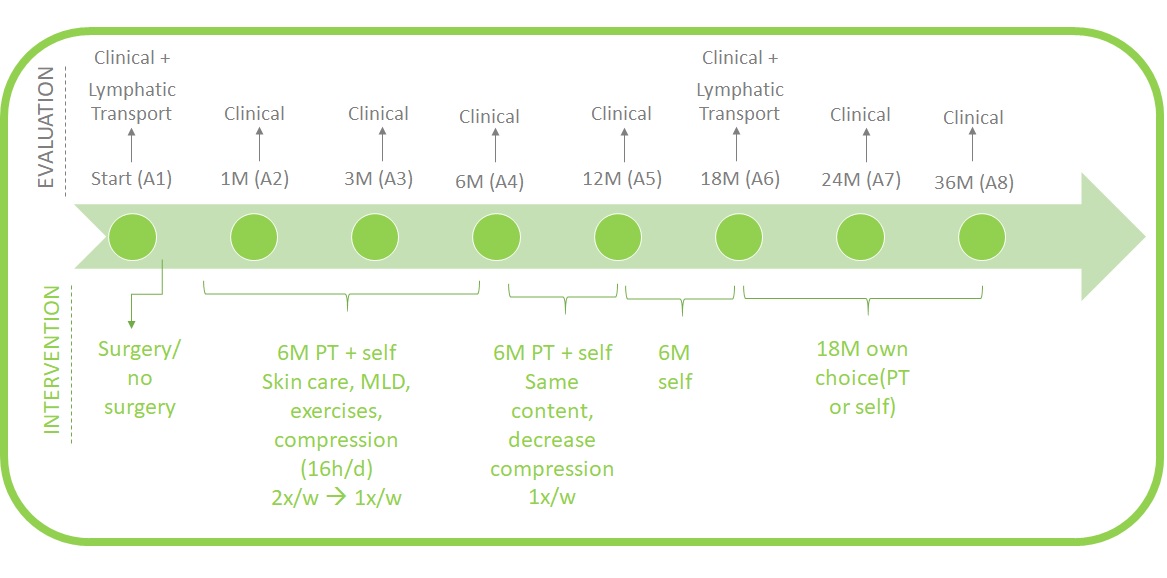
Oh yes, there are! In October 2021, the SurLym Trial will start including 180 patients with primary or secondary lymphedema at the upper and lower limb. This is a pragmatic multicenter randomized controlled trial approved by the Ethical Committees of the main and participating centers.
Aim: To investigate the added value of reconstructive lymphatic surgery to the conservative
treatment in patients with lymphedema
Treatment:
Included patients will be randomized in either the intervention or either the control group:
- Intervention group: reconstructive surgery (LVA and/or LNT depending on the patient’s individual situation) by experts followed by usual care (DLT) during 18 months
- Control group: no surgery, only usual care (DLT) during 18 months
Follow-up:
Both groups will receive a regular follow-up until 36 months after the start of the trial. On regular time points there will be an evaluation of the clinical outcomes and the treatment of lymphedema by a therapist. There are also some technical investigations at the start and after 18 months, like a lymphofluoroscopy and a lymphoscintigraphy. In case of LVA, a lymphatic MRI will also be conducted. In case of LNT, a CT angiography will be performed.
Who can participate:
- Objective diagnosis of primary or secondary lymphedema stage I, IIa or IIb at the upper or lower limb
- In case of cancer-related lymphedema: approval for participation from oncologist
- History of at least 6 months of DLT until minimal pitting
- >18 years old
- Not pregnant and no wish of becoming pregnant during first 18 months of follow-up
- No history of LVA or LNT of liposuction
- BMI<35
- No presence of chronic venous insufficiency C4, C5, C6 or deep venous thrombosis or post-thrombotic syndrome (in case of lower limb lymphedema)
- No allergy for Indocyanine green, iodine, penicillin or sulphonamides.
- No increased activity of thyroid gland or a benign tumor of the thyroid gland, no severe renal insufficiency and no heparin use.
Where: UZ Leuven, UZ Gent or CHU UCL Mont-Godinne
More information:
https://kce.fgov.be/nl/kce-191245-vergelijking-van-reconstructieve-chirurgie-versus-geen-chirurgie-additioneel-aan-de (more information will appear soon)
or you can send an e-mail with your questions to studiechirurgielymfoedeem@uzleuven.be
An-Kathleen Heroes
References
- Damstra, Robert J, et al. The Dutch Lymphedema Guidelines Based on the International Classification of Functioning, Disability, and Health and the Chronic Care Model. Journal of Vascular Surgery. Venous and Lymphatic Disorders (New York, NY), vol. 5, no. 5, 2017, pp. 756–765.
- ISL. The Diagnosis and Treatment of Peripheral Lymphedema: 2020 Consensus Document of the International Society of Lymphology. Lymphology 2020; 53(1):3-19.
- Nielubowicz J, Olszewski W. Surgical lymphovenous shunts for decompression of secondary lymphoedema. J Cardiovasc Surg (Torino) 1966;7:262–267.
- Jacobson JH 2nd, Suarez E L. Microvascular surgery. Dis Chest. 1962 Feb;41:220-4.
- Laine J B, Howard J M. Experimental lymphatico-venous anastomosis. Surg Forum. 1963;14:111–112.
- Sedlácek J. Lymphovenous shunt as supplementary treatment of elephantiasis of lower limbs. Acta Chir Plast. 1969;11(2):157-62.
- Koshima I, Nanba Y, Tsutsui T, Takahashi Y, Itoh S. Long-term follow-up after lymphaticovenular anastomosis for lymphedema in the leg. J Reconstr Microsurg. 2003 May;19(4):209-15.
- O'Brien BM, Chait LA, Hurwitz PJ. Microlymphatic surgery. Orthop Clin North Am. 1977 Apr;8(2):405-24.
- Kung TA, Champaneria MC, Maki JH, Neligan PC. Current Concepts in the Surgical Management of Lymphedema. Plast Reconstr Surg. 2017 Apr;139(4):1003e-1013e.
- Clodius L, Smith PJ, Bruna J, Serafin D. The lymphatics of the groin flap. Ann Plast Surg. 1982 Dec;9(6):447-58.
- Chang EI, Chu CK, Hanson SE, Selber JC, Hanasono MM, Schaverien MV. Comprehensive Overview of Available Donor Sites for Vascularized Lymph Node Transfer. Plast Reconstr Surg Glob Open. 2020 Mar 27;8(3):e2675.
- Visuri MT, Honkonen KM, Hartiala P, Tervala TV, Halonen PJ, Junkkari H, Knuutinen N, Ylä-Herttuala S, Alitalo KK, Saarikko AM. VEGF-C and VEGF-C156S in the pro-lymphangiogenic growth factor therapy of lymphedema: a large animal study. Angiogenesis. 2015 Jul;18(3):313-26.
- Alitalo K, Tammela T, Petrova TV. Lymphangiogenesis in development and human disease. Nature. 2005 Dec 15;438(7070):946-53.
- Lin CH, Ali R, Chen SC, Wallace C, Chang YC, Chen HC, Cheng MH. Vascularized groin lymph node transfer using the wrist as a recipient site for management of postmastectomy upper extremity lymphedema. Plast Reconstr Surg. 2009 Apr;123(4):1265-1275.
- Scaglioni MF, Fontein DBY, Arvanitakis M, Giovanoli P. Systematic review of lymphovenous anastomosis (LVA) for the treatment of lymphedema. Microsurgery. 2017 Nov;37(8):947-953.
- Rosian K, Stanak M. Efficacy and safety assessment of lymphovenous anastomosis in patients with primary and secondary lymphoedema: A systematic review of prospective evidence. Microsurgery. 2019 Nov;39(8):763-772.
- Forte AJ, Khan N, Huayllani MT, et al. Lymphaticovenous Anastomosis for Lower Extremity Lymphedema: A Systematic Review. Indian J Plast Surg. 2020;53(1):17-24.
- Park KE, Allam O, Chandler L, et al. Surgical management of lymphedema: a review of current literature. Gland Surg. 2020;9(2):503-511. doi:10.21037/gs.2020.03.14
- Dionyssiou D, Demiri E, Tsimponis A, Sarafis A, Mpalaris V, Tatsidou G, Arsos G. A randomized control study of treating secondary stage II breast cancer-related lymphoedema with free lymph node transfer. Breast Cancer Res Treat. 2016 Feb;156(1):73-9.
